Équipe Philippe MARIN
Neuroprotéomique et signalisation des pathologies cérébrales
Project Temperature and Circadian control of Alzheimer’s disease pathophysiology
PRINCIPAL INVESTIGATOR

IGF staff involved
Laurent GIVALOIS
CRCN CNRS
Sylvie CLAEYSEN
CRHC Inserm
Philippe MARIN
DRCE CNRS
Carine BÉCAMEL
PU UM
Charleine ZUSSY
Chercheur CDD UM
Magalie MATHIAS
IE CDD UM
Nicolas MARCHI
CRHC CNRS

Alzheimer’s disease (AD) is a progressive neurodegenerative disorder defined by β-amyloid (Aβ) plaques and tau tangles whose prion-like propagation parallels cognitive decline. Sleep and AD interact bidirectionally: sleep disruption elevates Aβ and tau levels and accelerates their spread. Under physiological conditions, the sleep–wake cycle (SWC) regulates Aβ and tau levels through secretion, neuronal reuptake, and glymphatic clearance. In the same line of evidence, the SWC-variation in body temperature (BT) is blunted in AD and has been shown to worse pathology. Our experimental data indicate BT fluctuations drive tau proteostasis: lower BT during sleep increases tau phosphorylation and suppresses its secretion. This project will test how BT modulates Aβ/tau reuptake, clearance, and propagation and evaluate thermal therapy as a non-pharmacological complementary approach.
We hypothesize that circadian BT variations regulate Aβ and tau secretion, reuptake and clearance. This project aims to unravel the vicious cycle linking thermoregulatory deficits, sleep disturbances, and glymphatic dysfunction to the propagation of Aβ and tau. Understanding these connections will enable to propose innovative therapeutic strategies. The following specific objectives will be pursued:
- Aim 1. Determine whether sleep–wake body temperature (BT) variation regulate tau reuptake.
Define how sleep–wake BT fluctuation (35–38°C) regulate neuronal reuptake and cell-to-cell propagation of tau (role of Syndecan-3). - Aim 2. Determine whether sleep–wake BT variations regulate Aβ propagation.
Determine how BT modulates Aβ production and reuptake (role of Syndecan-3), and its propagation using in vitro, in vivo, and clinical correlative readouts. - Aim 3. Test whether genetic inhibition of SDC3 prevents the propagation of amyloid and tau pathologies in mouse models of AD subjected to sleep deprivation.
Test whether genetic SDC3 inhibition (KO/siRNA) blocks tau and Aβ secretion/uptake/propagation and rescues cognition in mice exposed to chronic sleep deprivation; quantify ISF/CSF tau/Aβ and pathology. - Aim 4. Determine whether BT influences the glymphatic clearance of Aβ and tau.
Define how BT modulates glymphatic clearance of tau/Aβ (AQP4 expression/polarization, microdialysis ISF↔CSF/plasma flux). Evaluate controlled heat exposures as a non-pharmacological strategy, and probe consequences of AQP4 inhibition on sleep, thermoregulation, and pathology.
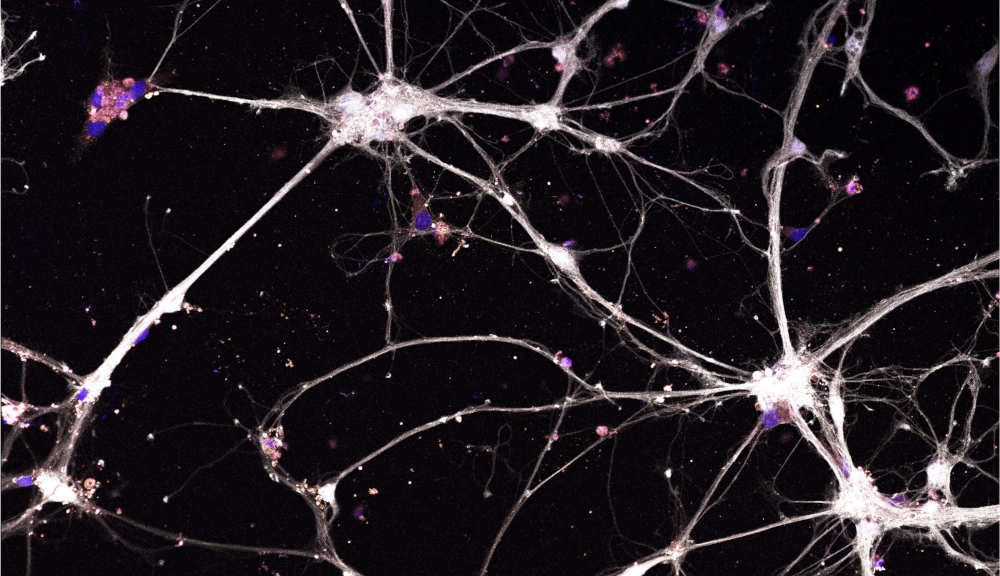
Confocal image of primary mouse cortical neurons stained for red fluorescent protein (RFP, white), DAPI (blue), syndecan-3 (purple). Cells were exposed at 38°C for 72 hours. 20X magnification.
Main publications
• Canet G., et al. (2025) J Clin Invest, 135, e182931.
• Canet G., et al. (2025) Brain, 148, 9-11.
• Canet G., et al. (2023) Exp Neurobiol, 32, 423-440.
• Guisle I., et al. (2022) Neurobiol Aging, 113,118-130.
Funding
• 2026-2029 Association France Alzheimer – ARCP
Collaborations
• Emmanuel Planel (Quebec, QC, Canada)
• Esther M Blessing (New York, NY, USA)
• Brendan P Lucey (St. Louis, MO, USA)


