For nearly 20 years, it has been suggested that the activity of membrane receptors can be regulated by other receptors which, through direct interaction, stabilize an active or inactive conformation of their target. This hypothesis offers cells the possibility of integrating external signals that act on the multitude of receptors on their surface. Although appealing, this hypothesis has often been questioned and, in any case, has never been clearly illustrated.
Through collaboration between the teams of Philippe Rondard at the IGF, Jianfeng Liu in Wuhan, and Jesús Giraldo in Barcelona, it has recently been shown how a class A receptor can stabilize the mGlu2 glutamate receptor in its active form.
The mGlu2 receptor is a constitutive dimeric receptor in which glutamate binding causes significant relative movement between the two subunits. This movement is such that the contact interface at the membrane domains is completely different between the inactive form (which involves transmembrane helices 3 and 4) and the active form (which involves transmembrane helices 6). The authors show that by selectively interacting with the 3-4 interface of the mGlu2 recexptor, which is inaccessible when the receptor is inactive, the serotonin 5HT2a receptor stabilizes the mGlu2 receptor in its active form. These results, based on complementary biochemical approaches and the use of different biosensors, demonstrate that a membrane receptor can act as a positive allosteric modulator of an mGlu receptor. The authors also identify several receptors capable of controlling mGlu2 activity in this way.
Given that mGlu2 and 5HT2a receptors are targets for antipsychotic drugs, a better understanding of their functional interaction could open up new avenues for treating schizophrenia more effectively.
Other mGlu receptors, such as mGlu5, are also recognized as targets of G protein-coupled receptors, as are dopamine and adenosine receptors. A similar regulatory mechanism is highly likely, as already illustrated in work carried out in collaboration with Julie Perroy’s team at the IGF (Sebastianutto et al., J Clin Invest, 2020), showing the importance of such a phenomenon in Parkinson’s disease.
This work has just been published in the journal Nature Communications.
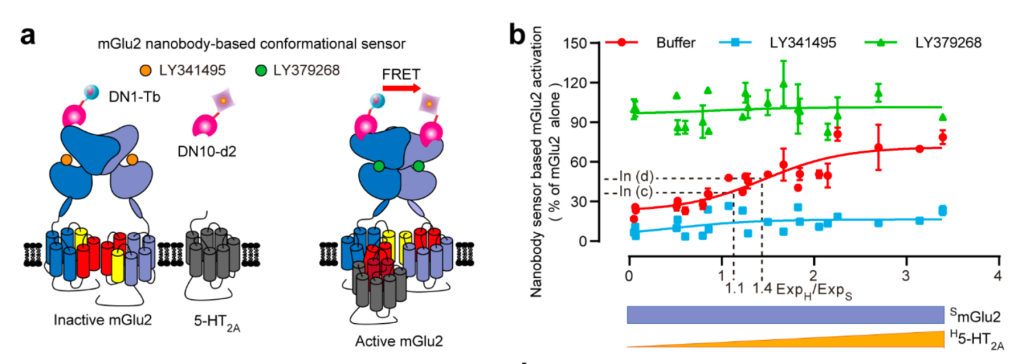
The serotonin 5HT2a receptor, by interacting specifically with the active form of the mGlu2 receptor, is able to stabilize it in its active form. a) Diagram illustrating the interaction of the 5HT2a receptor with the transmembrane helices of the mGlu2 subunits, which are only available when the receptor is active (shown in red). The active form of the receptor is detected using two nanobodies, one of which binds only to the active form, the only situation that generates a FRET-type energy transfer signal. b) Change in the proportion of mGlu2 receptors in the active form detected using the two nanobodies in the presence of increasing amounts of the 5HT2a receptor. In the presence of the mGlu2 agonist, shown in green, the receptors are always in their active form (high FRET), whereas in the presence of the antagonist, shown in blue, the receptors remain in their inactive form (low FRET). In the absence of the mGlu2 ligand, the proportion of active receptors increases with the amount of 5HT2a receptors.


