The GABAB receptor was the first mandatory heterodimeric receptor identified (1998), being composed of two similar subunits encoded by two distinct genes: GABAB1 and GABAB2. Structural analysis revealed that each subunit has a specific role, the GABAB1 subunit contains the agonist binding site, while the GABAB2 subunit is responsible for G protein activation. How ligand binding in one subunit can lead to the activation of the associated subunit has been much studied by the Jean-Philippe Pin’s group at IGF, leading to the proposal that agonist binding in the GABAB1 binding domain induces its closure, resulting in a relative movement between the subunits, leading to a new active interface of their membrane domain involving the transmembrane helix 6. Such proposal was formally validated through the cryoEM structure determination of the GABAB receptor in its basal and active state, with and without the associated G protein. These structural studies revealed a very different mode of G protein activation than that used by most other GPCRs, with no movement of TM6, and a G protein interaction on the side of the GABAB2 subunit, involving the intracellular loops. This mechanism was then observed for all class C GPCRs.
The GABAB receptor is structurally more complex than most other GPCRs. The group led by Jean-Philippe Pin has demonstrated, thanks to the development of the SNAP htrf technology in collaboration with Revvity, that i) the GABAB receptor could associate into larger entities, composed at least of two GABAB heterodimers, associated via the GABAB1 subunit, and ii) such complexes are stable at the cell surface, and lead to a negative allostery between the heterodimers, as only one can bind a ligand and signal. Several lines of evidence support the presence of GABAB tetramers in the brain, including proteomic analysis, and proximity assays using subunit specific antibodies. They even support that most GABAB receptors are indeed in a multimeric quaternary organization in native cells. Through the collaboration between the team « Neuroreceptors, dynamics and functions » headed by Philippe Rondard at IGF, the Jianfeng Liu’s lab in Wuhan (China), and Yan Zhang’s lab in Hangzhou (China), the cryoEM structure of the GABAB tetramer was solved. This structure largely confirms previous data, and illustrates how one GABAB1 subunit prevents ligand binding in the associated one, through an asymmetric binding interaction between the GABAB1 binding domains. Although such a negative allostery between the heterodimers within such complexes questions their physiological relevance, some genetic variants associated with human diseases, mainly affect the function of the tetramers, rather than the heterodimers, pointing at the former as important players for normal brain functions. More work will be needed to better understand the physiological relevance of such a large quaternary structural organization of a GPCR.
Such findings open much novel possibilities to finely tune GABAB receptor activity in the brain, and further illustrate how unique this receptor is.
This work has just been published in the journal Nature Communications.
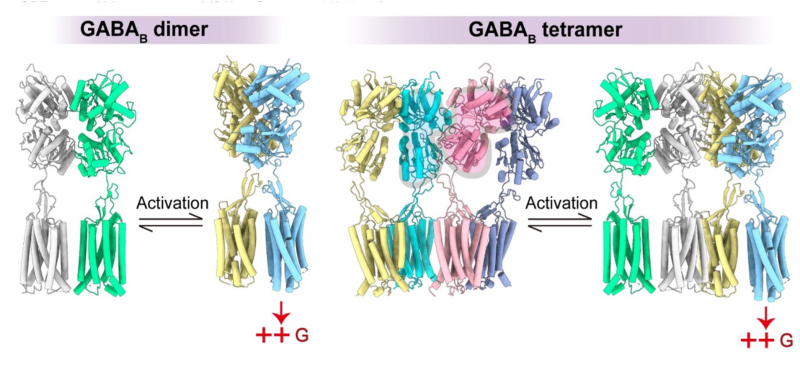
Different activation mechanisms for dimeric and tetrameric GABAB receptors. In the case of the dimer, GABA binding to the GB1 subunit (gray) leads to the closure of the VFT (yellow) and to the relative movement of the two subunits relative to each other, leading to the activation of the membrane domain of GB2 (blue), which can then activate a G protein. In the case of the tetramer, the asymmetric association of the two GB1 subunits (blue and pink) leads to the closure of a single GB1 (yellow), and consequently to the activation of a single GB2 (blue). But what could be the physiological significance of such a system? We do not have the answer to this question, but we can speculate that the second receptor within the tetramer allows for the action of interacting proteins and potentially the activation of other signaling pathways.


