Équipe Philippe MARIN
Neuroprotéomique et signalisation des pathologies cérébrales
Project HYDRA – Homeostasis and Physiological Dysregulation in Alzheimer’s Disease
PRINCIPAL INVESTIGATOR

IGF staff involved
Geoffrey CANET
CRCN, CNRS
Carine BÉCAMEL
PU, UM
Sylvie CLAEYSEN
CRHC, Inserm
Marie-Laure PARMENTIER
DR2, Inserm
Charleine ZUSSY
Chercheur CDD, UM
Maëlle COLLADANT
Doctorante, Inserm
Magalie MATHIAS
IE CDD, UM
Etienne AUDINAT
DR1, CNRS
Cyril GOUDET
DR2, CNRS
Nicolas MARCHI
CRCN, CNRS
Jérémie NAUDÉ
CRCN, CNRS
Tristan MOUJELIL-LEGAGNEUR
IE CDD, Inserm

The overall objective of HYDRA is to characterize the impact of exogenous disturbances (such as chronic stress, epilepsy, temperature variations, etc.) on the pathophysiology of Alzheimer’s disease (AD) and to develop innovative therapeutic strategies (such as the use of selective modulators of glucocorticoid receptors, multi-target molecules, or photopharmacology on the mGlu5 glutamate receptor) to counteract the counteract the progression of the pathology.
This project is divided into four main areas:
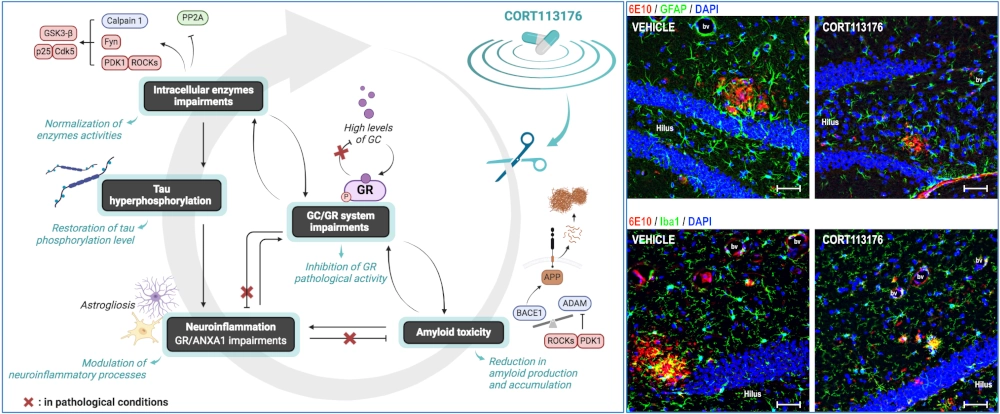
Stress, Glucocorticoids & AD
In sporadic forms of AD (more than 99% of patients), although the mechanisms remain poorly understood, certain risk factors have been identified. The primary risk factor is aging, but it is increasingly evident that certain life events, such as chronic stress or stress-related conditions like depression, can increase the likelihood of developing this neurodegenerative disease.
Thus, the main objectives of this preclinical project based on in vivo studies are:
- To confirm that chronic stress is a significant risk factor in the onset and/or progression of AD.
- To precisely dissect the role of the hypothalamic-pituitary-adrenal (HPA) axis (stress axis), glucocorticoids (GC), and their receptors (GR) in the etiology of AD.
- To evaluate the therapeutic potential of selective GR modulators (in collaboration with the U.S. company Corcept Therapeutics) to combat stress-related disorders and thereby limit or delay the progression of AD.
Main publications
• Canet G et al. (2025) Br J Pharmacol, 182,1930-1956.
• Canet G et al. (2022) Neuroendocrinology, 112, 982-997.
• Canet G et al. (2020) FASEB J, 34, 1150-1168.
• Canet G et al. (2019) Front Aging Neurosci, 11, 269.
• Canet G et al. (2018) Front Neurosci, 12;739.
• Pineau F et al. (2016) Neurobiol Aging, 45, 109-22.
• Brureau A et al. (2013) Neurobiol Aging, 34, 1426-1439.
Funding
• 2019 – MUSE – Explore#2 – Coordinator
• 2017-2018 – France Alzheimer – Projet GrAlz – Coordinator
Collaborations
• Hazel Hunt (Menlo Park, USA)
• Onno Meijer (Leiden, Netherland)
• Emmanuel Planel (Quebec, Canada)
Alumni
• Mathieu Vitalis (Engineer, 2020-2024)
• Célia Hernandez (PhD, 2018-2020)
• Geoffrey Canet (PhD, 2016-2020)
• Anthony Brureau (PhD, 2007-2010)
• Charleine Zussy (PhD, 2006-2009)
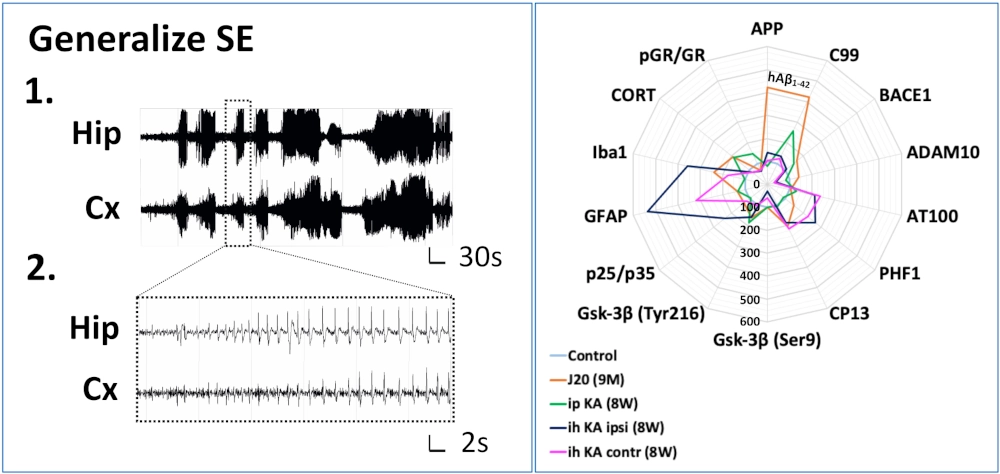
Epilepsy & AD
Several epidemiological studies have shown that epilepsy appears to impact the onset and/or progression of certain neurodegenerative diseases, particularly AD, and could therefore be considered a risk factor. This risk factor may vary depending on the age of onset of the first epileptic seizure.
We have conducted preliminary experiments on young animals using two mouse models of epilepsy (intra-hippocampal and intraperitoneal kainate injections). These experiments have revealed the long-term induction of AD-associated markers and identified shared pathways between the two pathologies.
The goal of this project is to assess the impact of the age of onset of a single generalized seizure in animals of different ages, with or without an Alzheimer-like phenotype (J20 transgenic mice vs. WT mice), and to evaluate the therapeutic potential of selective GR modulators in preventing epilepsy-associated neuroinflammatory processes.
Main publications
• Janvier A et al. (2025) Neurobiol Dis 217, 107177.
• Cresto N et al. (2025) Trends Mol Med, 31, 535-547.
• Canet G et al. (2022) Epilepsia, 63, 919-935.
• Zub E et al. (2019) FASEB J, 33, 13998-14009.
Funding
• 2022-2026 – ANR – Projet EpiNeurAge – Co-coordinator
Collaborations
• Emmanuel Planel (Quebec, Canada)
Alumni
• Mathieu Vitalis (Engineer, 2020-2024)
• Célia Hernandez (PhD, 2018-2020)
• Geoffrey Canet (PhD, 2016-2020)
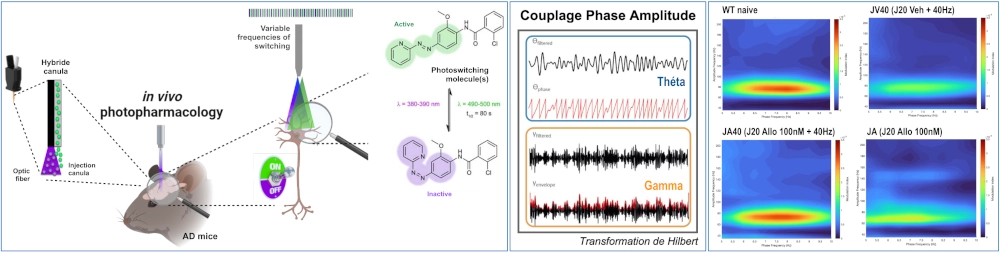
Oscillatory Neuronal Activity, mGlu5 & AD
Brain function is characterized by oscillatory neuronal activity, which plays a crucial role in processes such as memory and sleep. Recently, AD has been conceptualized as an “oscillopathy”, as an increasing number of studies on both AD patients and animal models report significant alterations in neuronal oscillatory activity. These alterations, occurring across a broad frequency spectrum, may contribute to memory deficits, sleep disturbances and reduced clearance of β-amyloid (Aβ) peptides, promoting their accumulation in the brain.
One of the main neurotransmitters involved in oscillatory activity, which is highly disrupted in AD, is glutamate. Therefore, this project focuses specifically on glutamatergic neurons in the hippocampus.
Photopharmacology is an innovative experimental strategy that uses light to control endogenous modulatory systems, such as metabotropic glutamate receptors (mGluR). Thus, our goal is to use photoswitchable ligands for mGluR to modulate in vivo oscillatory neuronal activity in mouse models of AD, ultimately aiming to counteract memory and metabolic deficits associated with the disease.
Main publications
• Zussy C et al. (2025) BioRxiv (doi : 10.1101/2025.10.03.680254).
• Dumazer A et al. (2025) Biochem Pharmacol, 240, 117065.
• Notartomaso S et al. (2024) Elife 13.e94931.
• Dumazer A et al. (2024) ACS Chem Neurosci, 15, 645-655.
Funding
• 2020-2024 Univ. Montpellier – MUSE AAP2020 – Projet GAiA – Coordinator
• 2021-2024 Fondation Recherche Médicale (FRM) Neurodegenerative Diseases AAP2021 – Projet OPA – Coordinator
• 2025 FRM Prematuration AAP2024 – Coordinator
• 2025 Caixa Fundation (Spain) – Prematuration – Partner
• 2025-2029 PEPR LUMA – HELIOPHORE Project– Partner
Collaborations
• Amadeu Llebaria (Barcelona, Spain)
• Xavier Gomez-Santacana (Barcelona, Spain)
• Emmanuel Planel (Quebec, Canada)
• Romain HAUDECOEUR (Grenoble)
Alumni
• Mathieu Vitalis (Engineer, 2020-2024)
• Anaëlle Dumazer (PhD, 2020-2023)
Climate change & AD
In the context of climate change and, more broadly, the environmental crisis, population exposure to environmental risks is increasing, whether through direct exposure (extreme weather events, wildfires, etc.) or indirect exposure (information about pandemics, pollution, consequences of droughts, etc.), promoting higher levels of anxiety in the population, and in particular, eco-anxiety. In this context, the Nexus CLIMAnX project brings together 4 areas of expertise in biological and epidemiological sciences as well as in the humanities and social sciences. It will investigate the impact on mental health (levels of anxiety and depression, and for human populations, levels of eco-anxiety and perceived well-being) of a physical factor (heatwave episodes) experienced directly, as well as other indirect exposure factors.
Our objective in this multidisciplinary project is to study, in a rodent model, the short- and long-term impact of repeated exposure to heatwave temperatures on anxiety and depression levels, social behavior, sleep, and cognitive abilities, as well as the adaptive biological mechanisms involved and the consequences on neurodegenerative processes, particularly on key markers of AD.
Funding
• 2025-2029 – DOCTORAL NEXUS ExposUM 2025 – Projet CLIMAnx – Partenaire
Collaborations
• Manuel Bächtold and Laurent Brun (LIRDEFn Montpellier)
• Hélène Rey-Valette and Cécile Bazart (CEE-M, Montpellier)


