Llama mini-antibodies targeting glutamate receptors rescue behavioral deficits in pre-clinical models of schizophrenia.
Mental disorders constitute one of the public health areas with the fewest progress over the last 50 years. Despite some recent advances in the treatment of depression and schizophrenia, new therapeutic avenues still need to be explored. In the particular case of schizophrenia, which affects around 1% of the population and often appears in young adults, current treatments essentially target the dopamine and serotonin systems. While they reduce certain symptoms, they have very little effect on cognitive disorders, i.e. problems of concentration, memory or organization, which severely handicap patients’ daily lives.
Over the past twenty years, research into schizophrenia has focused on glutamate, a major chemical messenger in the brain. A particular type of glutamate receptor, called a metabotropic receptor, appears to be a key to improving cognitive impairment. Today, the majority of treatments under development are based on small chemical molecules. Although easy to produce, these have a number of drawbacks: they often have undesirable effects and accumulate in the environment. Clinical trials of small molecules targeting glutamate receptors in schizophrenia have failed due to side effects or lack of efficacy. Researchers are now exploring a more natural alternative: therapeutic antibodies. These proteins, produced naturally by our immune system, are highly specific, biodegradable, well tolerated and designed to defend our organism. Their main limitation is their difficulty in reaching the brain, protected by a highly selective biological barrier, the blood-brain barrier.
This is where llamas play a surprisingly important role. These animals possess a particular type of antibody from which it is possible to generate mini-antibodies or nanobodies around ten times smaller than conventional antibodies. Until recently, scientists believed that nanobodies, like antibodies, could not cross the brain barrier. This study proves otherwise.
At the Institut de Génomique Fonctionnelle de Montpellier (IGF, CNRS, INSERM, University of Montpellier), Drs. Philippe Rondard, Julie Kniazeff and Jean-Philippe Pin (team “Neuroreceptors, dynamics and functions”) ternationally recognized experts in molecular pharmacology, have designed a nanobody that specifically activates the mGlu2 receptor, a metabotropic glutamate receptor. This work was carried out in collaboration with Dr. Patrick Chames (CRCM, INSERM, CNRS, University of Marseille), whose team developed the nanobodies from the immunization of llamas. Injected peripherally, these nanobodies reach the brain and correct cognitive disorders in two preclinical models of schizophrenia. The first is based on neonatal administration of phencyclidine, a known hallucinogenic psychotropic favouring the development of schizophrenia, in collaboration with Drs. Carine Bécamel and Philippe Marin at the IGF (team “Neuroproteomics and signalling of brain disorders”). The second model, of genetic origin, was developed by Dr Amy Ramsey’s team at the University of Toronto (Canada). It corresponds to animals displaying behavior reminiscent of the clinical signs of schizophrenia, associated with a deficit in the expression of the NMDA receptor that is critical for synaptic plasticity.
The results obtained with these two schizophrenia models are impressive: the cognitive functions of treated animals improve markedly after a single injection of the nanobody, with a prolonged effect lasting over a week. A four-week sub-chronic treatment protocol with reduced doses confirms the persistence of benefits over time. Further analysis, carried out in collaboration with Dr Denis Servent (CEA, Saclay), revealed that nanobodies injected into the abdominal area were able to penetrate the brain.
This work demonstrates that nanobodies offer a promising alternative to current approaches: they are effective, targeted, biodegradable and, above all, able to reach the brain. Unlike small molecules, they have limited side effects and environmental impact; and compared with conventional antibodies, nanobodies penetrate the brain more easily.
This breakthrough marks a major turning point for neuropharmacology and opens the way to a new generation of treatments for schizophrenia, but also potentially for other psychiatric disorders. Three patents have already been filed to protect this promising innovation.
This work has just been published in the journal Nature.
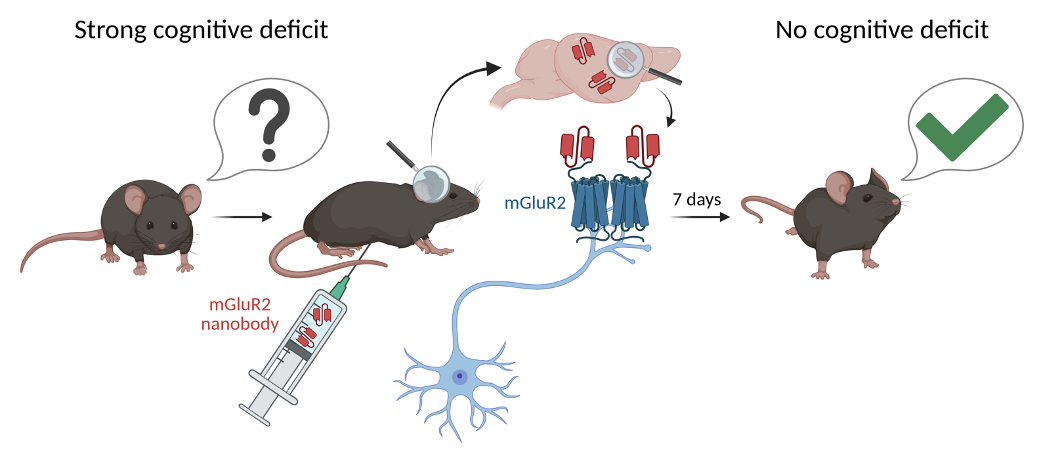
Schizophrenia is characterized by several phenotypes, including cognitive disorders, which are not corrected by current treatments. To fill this gap, we have developed a nanobody targeting the mGlu receptor which penetrates the brain after administration in the abdominal cavity and improves cognition in treated animals, an effect which lasts for up to 7 days.


