CARDIAC DEVELOPMENT, DISEASE AND REGENERATION
Department: Physiology and Cancer
Research subject
Our teams aims to decipher the mechanisms involved in cardiac development, disease and regeneration. Our research is divided into the following programs
The role of mechanosensitivity in cardiac development and disease.
Congenital heart defects (CHD) are an unfortunately common occurrence affecting upwards of 2% of all live births. The cardiac outflow tract (OFT) in humans is a transient structure which develops during gestation and will give rise to a number of arterial pole features such as the aortic valve. Hemodynamic forces are known to regulate cardiogenic events such as OFT development and valve formation. When these forces, or the sensory systems which detect them, are disrupted these developmental processes are also affected. Our goal is to define the signalling events downstream of hemodynamic forces that drive OFT/aortic valve development and to identify novel factors required for this process.
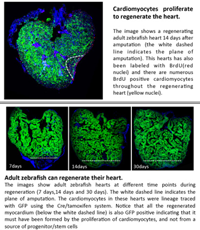
The role of mechanosensitivity in cardiac regeneration.
The loss of cardiac tissue following a heart attack results in drastic changes in mechanical loading. Cardiomyocytes respond to these changes by undergoing hypertrophy which will ultimately lead to heart failure. In contrast to adult mammals, neonatal mice and adult zebrafish can fully regenerate their hearts after an extensive insult. Changes in mechanical loading can be sensed by wide variety of mechanosensory mechanisms. Our goal is to identify the mechanosensors required to detect changes in mechanical loading and subsequently trigger cardiac regeneration.
The effect of environmental factors on Neuro/Cardiovascular development
The harmful effects of poisonous high-levels of environmental pollutants to the central nervous and cardiovascular systems are well known. We are testing the hypothesis that exposure to pesticide cocktails during development, in compositions and dosages pertinent to environmental contamination and to consumers, may result in neuro/cardiovascular defects.
Epigenetics of cardiac development and disease.
Little is known about the epigenetic regulation of cardiogenesis. It is now clear that DNA sequence alone does not underlie complex regulatory networks. The cohesin complex, has been shown to play a central role in the formation and abrogation of DNA loops. Patients with loss of function mutations in cohesin complex members (Cornelia de Lange Syndrome (CdLS)) present with defective cardiac development that is thought to result from widespread transcriptional dysfunction. Characterizing the defective chromatin conformational changes underlying these defects will yield novel insights into the epigenetic mechanisms governing cardiac development.
Team
Major publications
- Faucherre A, Nargeot J, Mangoni ME, Jopling C. piezo2b Regulates Vertebrate Light Touch Response.J Neurosci. 2013 Oct 23;33(43):17089-94. doi: 10.1523/JNEUROSCI.0522-13.2013. PMID: 24155313 [PubMed - in process]
- Faucherre A, Kissa K, Nargeot J, Mangoni M, Jopling C. Piezo1 plays a role in erythrocyte volume homeostasis.Haematologica. 2013 Jul 19. [Epub ahead of print]
- Jopling C, Suñe G, Faucherre A, Fabregat C, Belmonte-Izpisua JC. Hypoxia induces myocardial regeneration in zebrafish.Circulation. 2012 Nov 14.
- Faucherre A and Jopling C. The heart’s content-renewable resources.Int J Cardiol. 2012 Oct 5 S0167-5273(12)01157-6. Review.
- Jopling C, Sune G, Morera C, Belmonte JC. p38a MAPK regulates myocardial regeneration in zebrafish.Cell Cycle. 2012 Mar 11, 1-7.